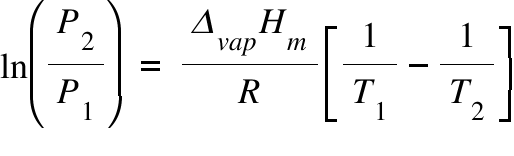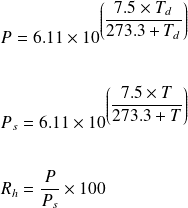Calculate vapour pressure of 0.1M urea soln. Vapour pressure of water at the given temperature is 20 torr. Assume molality and molality to be equal.

a) Vapour pressure of benzene is 200 mm of Hg. When 2 gram of a non-volatile solute dissolved in 78 gram benzene. Benzene has vapour pressure of 195 mm of Hg. Calculate

The relative lowering of vapour pressure of 1% Solution of Aniline in Ether was 0.007. Calculate the molecular weight of Aniline. - India Site

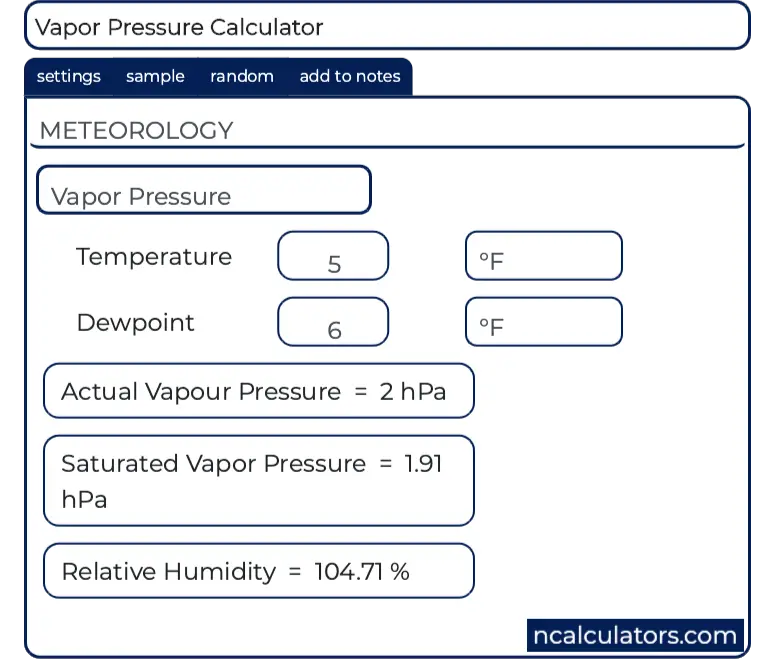
SOLVED: Over a water surface, the saturation vapor pressure (es) is related to the air temperature (T) by the following equation: 17.27T es = 611exp(237.3 + T) where es is in pascals
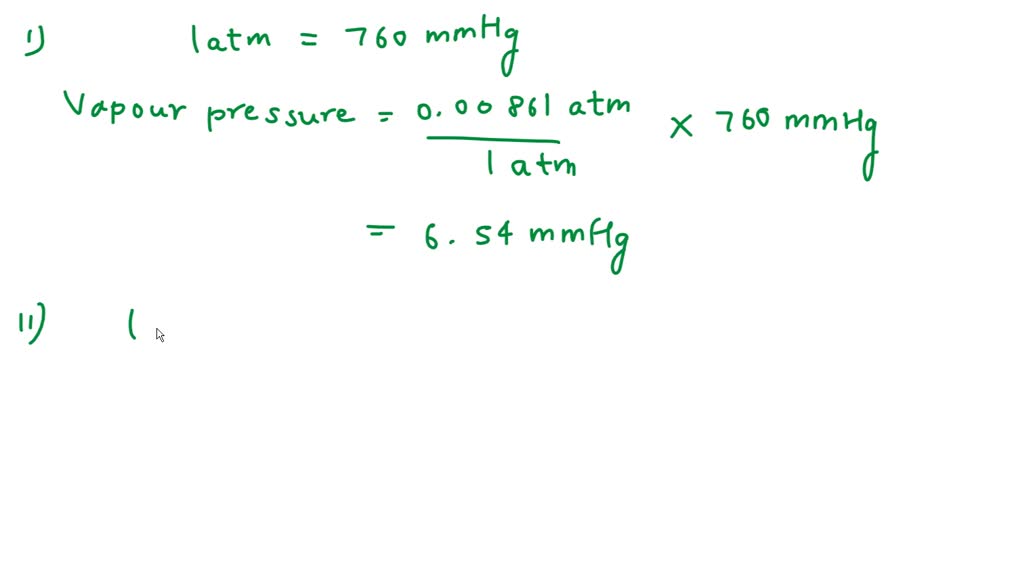
SOLVED: The vapor pressure of water at 5°C is 0.00861 atm. Calculate the vapor pressure in mmHg and torr. Round each of your answers to 3 significant digits. mmHg? torr?
The vapour pressure of pure water is 760 mm at 25^°C . The vapour pressure of solution containing 1(m) solution of glucose will be ??